Understanding Chemical Oxidation in 19th-Century Dyes
Oxidation: The Silent Color Killer
While UV degradation gets most of the attention in conservation literature, chemical oxidation is responsible for some of the most dramatic and difficult-to-predict color changes in 19th-century textiles. Unlike UV damage, which requires light exposure, oxidation happens in the dark. Textiles in storage, wrapped in acid-free tissue, sealed in archival boxes — they are still oxidizing, slowly but relentlessly.
For conservators, this means that even well-stored textiles can present color-matching challenges that differ fundamentally from UV-faded textiles. A textile that has been in dark storage for a century may show oxidation-driven color shifts with no UV fading at all, creating a degradation profile that does not match published UV-fading data.
How Oxidation Works on Organic Dyes
Oxidation in the context of textile dyes means the loss of electrons from the dye molecule, typically mediated by atmospheric oxygen. The process is accelerated by:
- Humidity — Water molecules facilitate the transfer of electrons between the dye and atmospheric oxygen
- Metal ion contaminants — Iron, copper, and manganese ions (from mordants, water, or environmental contamination) act as catalysts
- Acidic conditions — Acid hydrolysis weakens dye molecules, making them more susceptible to subsequent oxidation
- Heat — Higher temperatures increase the rate of all chemical reactions, including oxidation
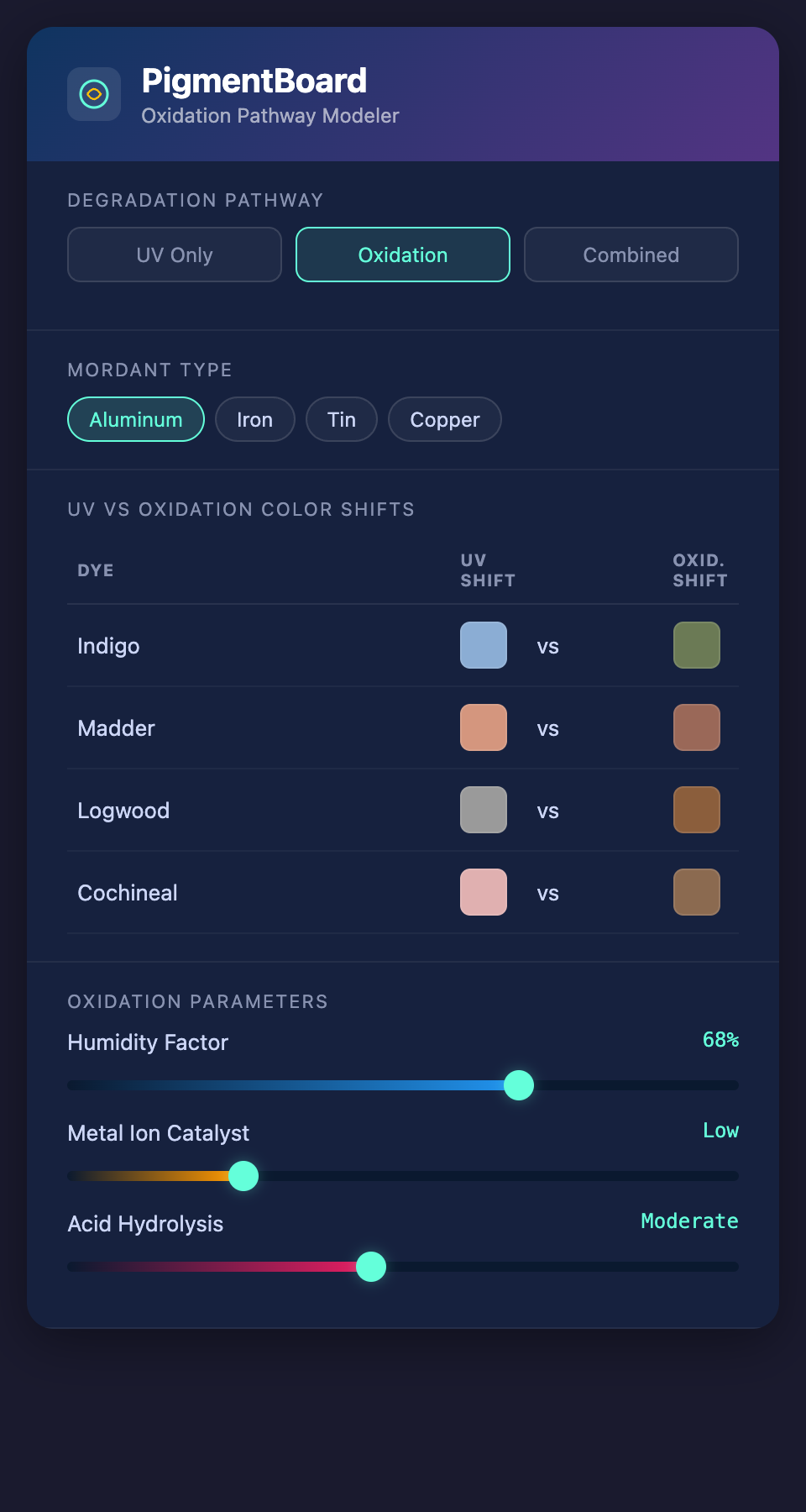
Specific Oxidation Effects on Common 19th-Century Dyes
Indigo undergoes one of the most well-documented oxidation pathways. The indigotin molecule (C16H10N2O2) oxidizes to isatin (C8H5NO2), which is yellow. This is why old indigo-dyed textiles often show a greenish or brownish cast — the blue of residual indigotin mixing with the yellow of isatin. In advanced oxidation, the textile can turn entirely brown.
Madder (alizarin and purpurin) oxidizes differently depending on the mordant used:
- With aluminum mordant: the red shifts toward a dull brownish-pink
- With tin mordant: the red shifts toward orange before fading
- With iron mordant: the already-dark shade deepens further toward near-black before eventually lightening
Logwood is among the most oxidation-sensitive dyes. Its primary colorant, hematoxylin, oxidizes to hematein (which is actually the desired purple-black dye compound), but further oxidation breaks hematein down into colorless fragments. This is why logwood blacks from the 19th century often show a characteristic rusty-brown shift — the dye has oxidized past its useful color state.
Cochineal (carminic acid) is moderately oxidation-resistant when properly mordanted with aluminum, but unmordanted or tin-mordanted cochineal oxidizes toward brown. The carminic acid molecule loses its anthraquinone chromophore through ring-opening reactions.
Turmeric and other curcuminoid dyes are extremely oxidation-sensitive. Curcumin oxidizes to vanillin and ferulic acid derivatives, both of which are colorless or pale yellow. Turmeric-dyed textiles from the 19th century rarely retain any visible color.
Oxidation vs. UV Degradation: Different Problems, Different Solutions
A critical distinction for conservators: oxidation and UV degradation produce different color shifts from the same starting pigment. This matters enormously when you are trying to match a faded color, because the correct match depends on identifying which degradation pathway — or which combination of pathways — caused the change.
| Dye | UV Degradation Shift | Oxidation Shift |
|---|---|---|
| Indigo | Lightens toward pale blue | Shifts toward brown-green |
| Madder (Al mordant) | Lightens toward pink-salmon | Shifts toward brownish-pink |
| Logwood | Lightens toward gray | Shifts toward rusty-brown |
| Cochineal | Lightens toward pale pink | Shifts toward brown |
When both UV and oxidation have acted on the same textile — which is common for textiles that were displayed and then stored — the resulting color is a compound of both pathways. Predicting this compound shift requires modeling both factors independently and combining them.
The Mordant Complication
Nineteenth-century dyers used metal salt mordants to fix dyes to fibers. These mordants do not just affect the original color — they change the degradation pathway. The same dye on the same fiber can age completely differently depending on the mordant:
- Aluminum mordants generally slow oxidation by stabilizing the dye-mordant complex
- Iron mordants accelerate oxidation by acting as catalysts (iron ions facilitate electron transfer)
- Tin mordants make fibers brittle and can accelerate both oxidation and UV degradation
- Copper mordants have complex effects — they can both stabilize certain dyes and catalyze oxidation of others
This means that a conservator examining a faded 19th-century textile needs to consider not just "what dye was used?" but "what dye was used with what mordant, and how does that mordant-dye combination oxidize?"
Practical Implications for Color Matching
Understanding oxidation pathways gives conservators a significant advantage in color matching:
1. Diagnose before you match. Before mixing any paint, determine whether the color change you see is primarily UV-driven, oxidation-driven, or a combination. This determines which degradation model to apply.
2. Look at protected areas. Areas hidden from light but still exposed to air (folds, seam allowances, areas under frames) show oxidation without UV damage. Comparing these to exposed areas helps separate the two factors.
3. Consider storage history. A textile that spent 80 years in a damp basement has a very different oxidation profile from one stored in a climate-controlled archive.
4. Adjust your formula for the dominant pathway. If oxidation is the primary driver, your target color will have more brown and less of the "bleached" quality that UV fading produces. Your pigment mixture needs to account for this.
Modeling Multiple Degradation Factors
The power of a multi-fader degradation model becomes clear when you consider oxidation alongside UV exposure. Rather than trying to intuit the combined effect of two (or more) chemical processes in your head, you can:
- Set the UV fader to the estimated light exposure
- Set the oxidation fader to reflect storage conditions and duration
- Set the humidity fader to influence the oxidation rate
- Set the mordant type to modify the degradation pathway
- Read the predicted color output
Each fader controls one variable. The model combines them according to published chemistry. The conservator evaluates the result and adjusts.
The Future of Oxidation-Informed Conservation
As we better understand the specific oxidation pathways of 19th-century dyes, conservation practice can move from reactive color-matching to proactive prediction. We can estimate how a textile will look in 50 years under given storage conditions. We can advise curators on which textiles are most at risk. And we can match existing degradation with a precision that trial-and-error mixing simply cannot achieve.
Ready to model oxidation, UV, and humidity together in one interface? Join the PigmentBoard waitlist and bring multi-factor degradation modeling to your lab.