Humidity's Hidden Role in Historic Pigment Degradation
The Overlooked Degradation Factor
Walk into any conservation lab and ask about pigment degradation, and the first response will almost always be about light. UV exposure, lux hours, light damage — these are the concepts that dominate conservation training and practice.
Humidity gets mentioned as an afterthought. "Keep it between 45-55% RH" is the standard advice, and for many conservators, that is where the conversation about moisture ends.
But humidity is not just a storage condition to be controlled. It is an active degradation agent that changes pigment chemistry in specific, predictable ways — ways that differ from UV damage and that must be accounted for separately when matching degraded colors.
How Humidity Degrades Pigments
Moisture affects pigments through several mechanisms, often working simultaneously:
1. Hydrolysis. Water molecules can directly break chemical bonds in dye molecules. This is especially significant for dyes that contain ester or glycosidic bonds — which includes many plant-based dyes. Hydrolysis fragments the dye molecule, reducing or eliminating its ability to absorb specific wavelengths of light.
2. Facilitated oxidation. Water acts as a medium for electron transfer between the dye molecule and atmospheric oxygen. In dry conditions, oxidation occurs slowly because the reactants have limited contact. In humid conditions, a thin film of water on fiber surfaces creates a reaction medium that dramatically accelerates oxidation rates.
3. Mordant dissolution. Metal mordants (aluminum, iron, tin) are bound to the fiber through coordination complexes. High humidity can partially dissolve these complexes, releasing the mordant and destabilizing the dye. Without its mordant, the dye loses both its color intensity and its resistance to further degradation.
4. Biological acceleration. High humidity promotes mold and bacterial growth, which produce enzymes and metabolic byproducts that attack dye molecules. The color changes from biological degradation are distinct from chemical degradation — often producing characteristic spotty patterns and musty, gray-brown discoloration.
5. Migration. Moisture can cause soluble dyes to migrate through the textile, creating halos, bleeding, and color redistribution that changes the apparent color of specific areas without changing the total amount of dye present.
Humidity-Specific Color Shifts
The color changes caused by humidity differ from UV-driven changes:
Madder reds in high humidity tend to shift toward a duller, muddier brown-red, as opposed to the pink-salmon shift caused by UV. This is because humidity-driven hydrolysis fragments the alizarin molecule differently than UV-driven photo-oxidation, producing different degradation products.
Indigo in high humidity can develop a greenish cast more rapidly than in dry conditions, because the water-facilitated oxidation of indigotin to isatin (yellow) occurs faster than the UV-driven pathway.
Iron-mordanted dyes in high humidity can produce localized darkening and black spotting, as moisture mobilizes iron ions which then catalyze aggressive oxidation of both the dye and the fiber.
Silk dyes in general are more humidity-sensitive than cotton or wool dyes, because silk's protein structure absorbs more moisture, creating a more reactive environment for the dyes.
The Interaction Between Humidity and UV
In real-world conditions, humidity and UV exposure act simultaneously, and their effects are not simply additive — they are synergistic. Humidity accelerates UV-driven degradation by providing water molecules that participate in the photochemical reactions.
Research has shown that:
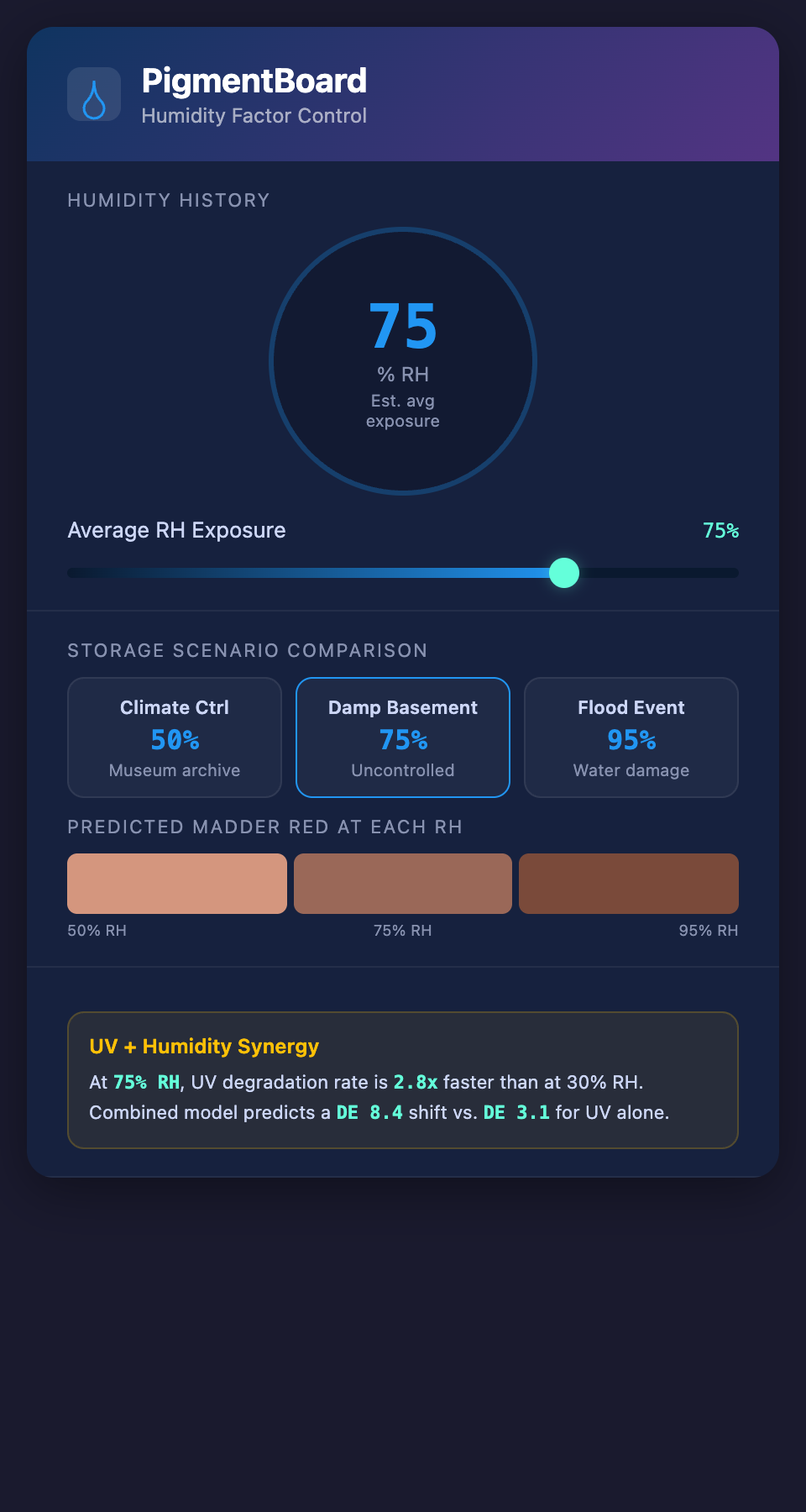
- A textile exposed to moderate UV at 75% RH degrades 2-4 times faster than the same textile at the same UV level at 30% RH
- The color shift direction can change depending on the humidity level during UV exposure
- Cycling humidity (alternating high and low) causes more damage than constant moderate humidity at the same average level
This synergy means that conservators cannot model UV degradation and humidity degradation independently and simply add the results. The two factors interact, and the interaction must be modeled as a combined system.
Practical Implications for Color Matching
1. Know the humidity history. Before matching a degraded pigment, determine — as closely as possible — the textile's moisture exposure history:
- Was it stored in a climate-controlled archive (45-55% RH) or an uncontrolled attic (potentially 80%+ in summer)?
- Was it displayed in a gallery with environmental controls or in a historic house with no climate management?
- Has it experienced flood damage, water stains, or high-humidity events?
2. Look for humidity-specific clues. Certain physical signs indicate humidity-driven degradation:
- Tide lines (water stain marks) showing dye migration
- Mold staining (gray, black, or greenish spots)
- Iron spot corrosion (dark spots around iron mordant areas)
- Fiber weakness or disintegration (especially in silk, where "shattering" is humidity-related)
3. Adjust your degradation model. If you are using a degradation model that only accounts for UV exposure, your predictions will be wrong for textiles with significant humidity exposure. You need a model that treats humidity as an independent variable — with its own fader, so to speak — that can be set independently of the UV factor.
4. Match under representative conditions. Humidity affects the appearance of pigments even in the present. A textile displayed in a gallery at 50% RH looks slightly different from the same textile at 40% RH, because moisture content affects the fiber's optical properties (translucency, surface reflection). Match your repair under conditions similar to the display environment.
Case Study: Two Identical Quilts, Two Different Fades
Consider two quilts made from the same fabric, by the same maker, in the same year (1865), using madder-dyed red cotton. One was kept in a well-ventilated New England farmhouse. The other spent decades in a damp Louisiana basement.
Despite having the same starting materials and approximately the same age:
- The New England quilt shows UV-dominant fading: the reds have shifted toward pale salmon-pink, with the strongest fading on the side that faced the window.
- The Louisiana quilt shows humidity-dominant degradation: the reds have shifted toward muddy brown-red, with the strongest changes in areas where the quilt was folded (trapping moisture) and near the floor (where humidity was highest).
A conservator matching both quilts would need two completely different formulas — even though the original color was identical. Without accounting for humidity as a separate degradation factor, the Louisiana quilt's color shift would be impossible to predict accurately.
Building Humidity Into Your Workflow
- Step 1: Assess the textile's humidity history as part of your initial examination
- Step 2: Note physical signs of humidity-specific degradation
- Step 3: Use a degradation model that includes humidity as an independent variable
- Step 4: Set the humidity parameter based on your assessment
- Step 5: Combine with UV and oxidation parameters for a comprehensive prediction
- Step 6: Fine-tune by comparing the model output to the physical sample
The Multi-Fader Advantage
This is precisely why a soundboard-style interface for degradation modeling is so powerful. Humidity, UV, oxidation, pollutants, biological agents — each one is a fader that can be adjusted independently. The model computes the combined effect, accounting for synergies and interactions. The conservator evaluates the result and adjusts.
No amount of visual intuition can compute the synergistic interaction between UV exposure at 300 klux-hours and humidity at 75% RH on tin-mordanted cochineal. But a well-built model can — and a soundboard interface makes it accessible without requiring a chemistry degree.
Ready to add humidity to your degradation modeling toolkit? Join the PigmentBoard waitlist and start modeling every factor that shapes historic pigment color.