Privacy and Consent in Medical-Informed Infant Memorial Content
The Problem: Memorials Built on Medical Data Without a Consent Architecture
A hospital bereavement program that pulls NICU chart data into a memorial record is not operating outside HIPAA — it is operating inside a very specific part of HIPAA that most programs do not read. The HHS guidance on health information of deceased individuals confirms that HIPAA's Privacy Rule protects the PHI of a deceased individual for 50 years after death. A detailed read of the rule at Bricker Graydon on HIPAA 164.502(f) spells out that uses and disclosures require authorization from the personal representative of the deceased — typically the parent for an infant.
A HIPAA Journal review of photography rules makes clear that photographs taken in a clinical setting are PHI and require written authorization to share, display, or move to a memorial system. The PMC best-practice bereavement photography analysis of 104 bereaved parents found that consent must be offered multiple times across the episode, because parents in acute shock often decline initially and later wish they had consented. A PMC paper on professional bereavement photography frames informed consent as an ethical mandate, not just a legal requirement. ACOG's Committee Opinion 786 on perinatal palliative care requires informed consent for life-limiting decisions, which includes memorialization decisions that flow from those clinical contexts.
The operational reality is that bereavement programs often rely on verbal assents captured by a nurse mid-shift, with no signed authorization, no revocation pathway, and no logging of which chart elements were extracted. This is legally precarious and ethically thin. The family deserves better, and the hospital's compliance posture needs stronger footing.
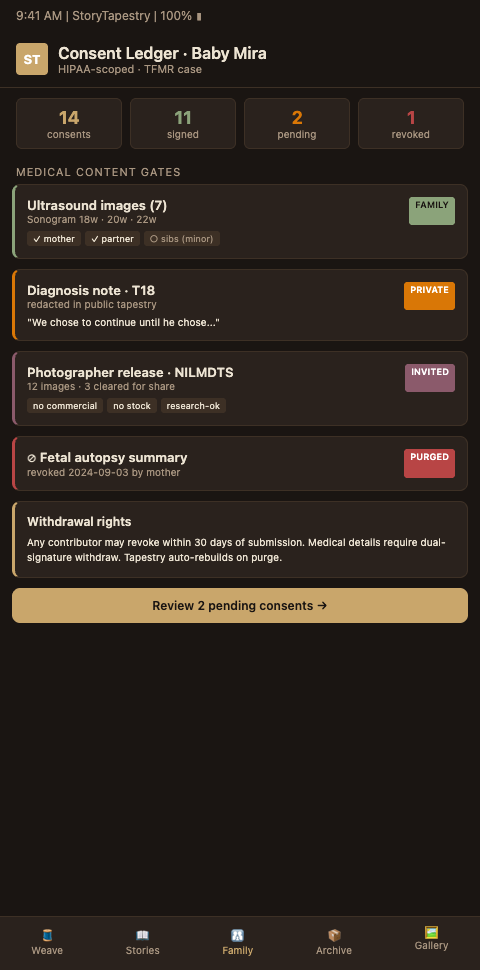
Solution Framework: Consent as a Tapestry Thread, Not a Form
StoryTapestry treats consent as an ongoing structural element of the memorial tapestry — a thread that runs alongside every other thread, versioned, revocable, and documented. A memorial is never built on implicit assent. Each medical-derived element has an explicit authorization trail, a visibility scope, and a revocation path.
The architecture starts with a layered consent flow. Hospital-based consent is offered in three phases. Phase one, at the point of loss or imminent loss, captures immediate wishes — photography yes/no, memory box yes/no, blessing/baptism offered. Phase two, 24-72 hours later, revisits the decisions with a rested parent and captures consent for medical-narrative extraction (which chart elements can become memorial threads), chaplain and social-worker involvement, and initial funeral-home coordination. Phase three, at 2-6 weeks post-loss, finalizes long-term consent — what persists in the tapestry, at what visibility level, with what sharing permissions. Each phase has its own authorization form, with separate signatures for each domain. The infant loss sensitivity practices shape how these phases are introduced, because a poorly-timed consent conversation harms more than a missing consent.
Granular scope per thread. Consent is not binary. StoryTapestry lets parents consent separately to: medical timeline extraction, photography, audio recordings, ultrasound preservation, clinician captions, sibling contributions, public community layer, Wave of Light participation, funeral-home sharing. Each scope has a signed record. Parents can decline some and accept others. This matches what the HIPAA Journal photography rule and the PMC professional-bereavement-photography paper both recommend: consent is specific, not blanket.
Revocation always available. A parent who consented at week two and then at month eighteen wishes to retract consent for a specific thread can do so through a one-click revocation inside the tapestry interface. StoryTapestry removes the thread from family view, updates its visibility state to "revoked," and logs the action. Revocation does not delete the underlying audit record — the hospital's compliance record requires a log of what was shared and when — but the thread no longer displays. The 50-year retention horizon under HIPAA means the audit record lives alongside the revocation, giving the hospital evidence of compliance without forcing continued display.
Personal-representative verification. For infant loss, the personal representatives are typically the parents, but state law varies on questions of unmarried parents, surrogate arrangements, and adoption. StoryTapestry captures personal-representative designation at intake and re-verifies at any escalation (e.g., when adding content that affects sibling visibility). The PMC professional-photography paper emphasizes the ethical weight of getting representative verification right, because the representative's authorization is what grants the program authority to operate on the baby's PHI.
Public vs private norms. Programs that offer communal observances — the Wave of Light wall, the Baby Loss Awareness Week shared thread — have to make the communal layer an explicit opt-in. Patterns developed for navigating public vs private norms across diaspora memorial contexts apply here: some families find community visibility healing, some find it intrusive, and the system's default must be the cautious one.
Cross-unit consent propagation. When OB bereavement coordination crosses L&D to NICU to postpartum, the consent record follows the tapestry rather than being rebuilt at each unit. A consent captured on L&D at phase one is honored on NICU at phase two without the family having to re-sign for the same scope. Each unit sees the current consent state and can add unit-specific scopes (a NICU nurse adding a skin-to-skin photograph gets the family's yes or no without re-asking about other scopes).
Advanced Tactics for Durable Privacy and Consent Operations
Four tactics strengthen a consent architecture past the baseline.
First, co-sign with the hospital compliance office. Draft the consent forms, retention policy, and revocation workflow with the compliance office as co-authors. A consent architecture produced by a bereavement program in isolation tends to lag HIPAA updates and state-law amendments. When compliance is on the author list, updates flow automatically into the memorial program's documentation.
Second, offer a privacy-education session as part of the family's onboarding. A 20-minute conversation with the bereavement coordinator covering what HIPAA protects, what the 50-year horizon means, what the family controls, and what the hospital controls, produces better long-term trust than a signed form the family never reads. The PMC best-practice photography paper documents that parents who understood consent deeply were measurably more satisfied at 6-month follow-up than parents who signed without comprehension.
Third, run an annual consent audit. Once a year, sample 20-30 active tapestries and verify: signed authorizations exist for each thread category, revocation logs are intact, personal-representative records are current, visibility scopes match signed scopes. Audit results feed back into staff training and form revision. Programs that do not audit drift toward non-compliance silently.
Fourth, plan for data subject rights under state privacy laws. California's CCPA, Virginia's VCDPA, Texas's DPSA, and a growing list of state privacy laws grant data-subject rights that interact with HIPAA in edge cases. Families asking "what do you have about my baby?" should receive a clear answer generated from the tapestry's access log. StoryTapestry provides a data-subject-access report that a coordinator can generate in five minutes.
Finally, document your child-of-deceased standard. When a deceased infant has a surviving sibling who at age 21 wants access to the memorial, the legal pathway is state-dependent. A pre-documented standard (what rights transfer, with what authorization) prevents a crisis at the moment of the sibling's first request.
CTA: For Hospital Bereavement Programs Tightening Privacy Architecture
Your current consent process probably relies on one or two forms, verbal nurse-captured assents, and an assumption that the rest is covered by general HIPAA compliance. StoryTapestry gives your program a layered consent flow, granular thread-level scopes, revocation pathways, and compliance-auditable logging. Schedule a 60-minute joint session with your bereavement coordinator and a member of the compliance office. We'll map your current consent forms against the HIPAA, ACOG, and state-privacy requirements and show the thread-level consent architecture side by side with your current workflow. The session covers the layered consent flow, the thread-level scope mapping, the revocation pathway library, the audit log specification, and a walkthrough of a sample consent matrix covering photography, clinical staff reflection, sibling participation, and sonography integration.
Pilot engagements include platform access for your bereavement coordinator and a compliance officer, a training block for your L&D and NICU teams on the layered consent workflow, and a named implementation lead who audits your first 10 family engagements for consent fidelity. Most programs complete consent architecture configuration within six weeks of contract signing and begin onboarding families the week configuration closes. Bring your bereavement coordinator, compliance officer, general counsel representative, and one L&D nurse manager — the session produces a compliance-ready consent workflow document the four of them can submit for institutional review before the pilot launches.